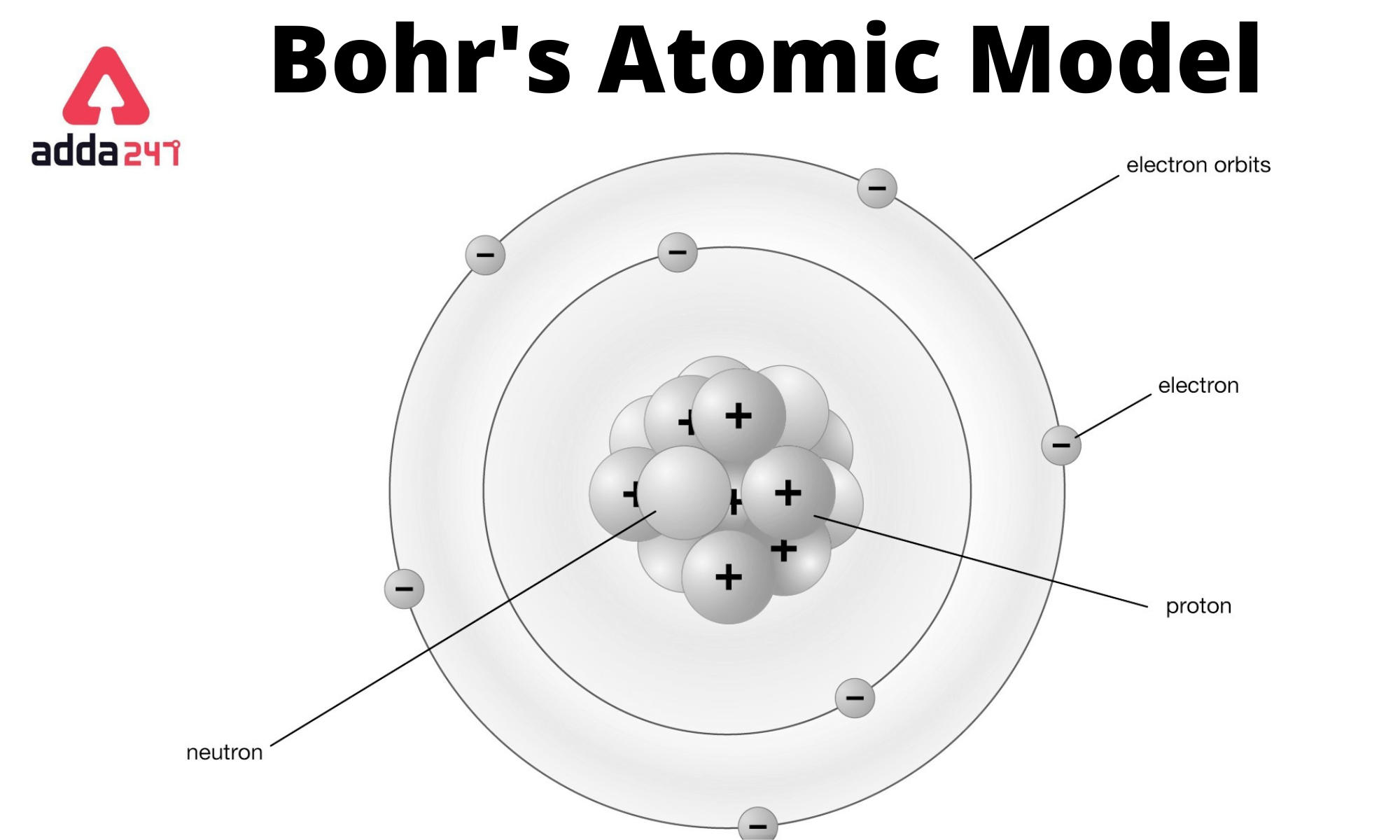
However, his model worked well as an explanation for the emissions of the hydrogen atom, but was seriously limited when applied to other atoms. 'blocks' in the periodic table, ie each row of transitional metals are filling a d shell, the first 3d, second 4d. Moving up the ladder increases your potential energy, while moving down the ladder decreases your energy.īohr's work had a strong influence on our modern understanding of the inner workings of the atom. The wave model explains atomic shell filling, the solving gives several types of orbitals, each with different allowed electrons, the s shell with 2, the p shell with 6, the shell with 10 and the f shell with 14. As you move up or down a ladder, you can only occupy specific rungs and cannot be in the spaces in between rungs. An everyday analogy to the Bohr model is the rungs of a ladder. The electron is not allowed to occupy any of the spaces in between the orbits. It is unable to explain the shape of the molecule. The orbits that are further from the nucleus are all of successively greater energy. Bohr model, description of the structure of atoms proposed in 1913 by the Danish physicist Niels Bohr. Bohr’s Atomic Model failed to explain the matter of the Stark effect when the spectral line from the atom during excitation gets split up into fine lines in the presence of a strong electric field. The ground state of the hydrogen atom, where its energy is lowest, is when the electron is in the orbit that is closest to the nucleus. Bohr’s model consists of a small nucleus (positively charged) surrounded by negative electrons moving around the nucleus in orbits. When the electron is in one of these orbits, its energy is fixed. It accounts for a wide range of physical phenomena, including the existence of discrete packets of energy and matter, the uncertainty principle, and the exclusion principle.Īccording to the Bohr model, often referred to as a planetary model, the electrons encircle the nucleus of the atom in specific allowable paths called orbits. Bohr’s solution was to incorporate Max Planck’s theory of radiation, which postulates that the energy radiation from an atomic system does not take place in the continuous way assumed in ordinary electrodynamics, but that it, on the contrary, takes place in distinctly separated emissions (Bohr 1913, p. This is a theory based on the principle that matter and energy have the properties of both particles and waves. Bohrs first postulate was that an electron in an atom could revolve in. This was the basis for what later became known as quantum theory. The Bohr model depicts the atom as a small, positively charged nucleus surrounded by electrons that travel in circular orbits around the nucleus similar in structure to the solar system, but with attraction provided by electrostatic forces rather than gravity. When the energy is removed, the electrons return back to their ground state, emitting a corresponding amount of energy-a quantum of light, or photon. 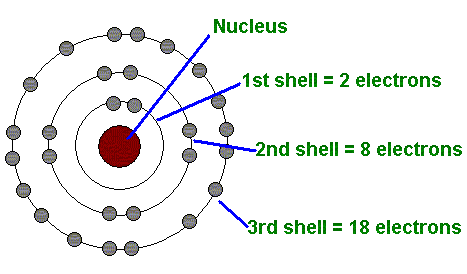
(Credit: Zachary Wilson Source: CK-12 Foundation License: CC BY-NC 3.0(opens in new window))īohr explained that electrons can be moved into different orbits with the addition of energy. \): Bohr's atomic model hydrogen emission spectra.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
